Inferior OMI ECG: Hyperacute T-Waves, Delayed Recognition, and Reperfusion Patterns
The 4 AM ECG of a chest pain patient in a community hospital
Written by Robert Herman, MD, PhD and Edited by Stephen W Smith (founder of Dr. Smith’s ECG Blog).
It’s 4 AM. A man in his 60s presents to a community hospital — no cath lab on site — with a history that should make any clinician sit up straight.
For the past two days: oppressive chest pain radiating to the throat, lasting under 10 minutes and then spontaneously resolving. The next day, symptom-free. Then, toward evening of day 2: the same pressure, same radiation, now coming in waves and not improving. Not provoked, not respiratory, not positional — though notably better sitting, worse lying down. No recent illness. No cardiac history documented.
He arrives. He looks okay. Alert, not clammy, well-perfused, sats 100%, no lung findings, muffled heart sounds but no murmur.
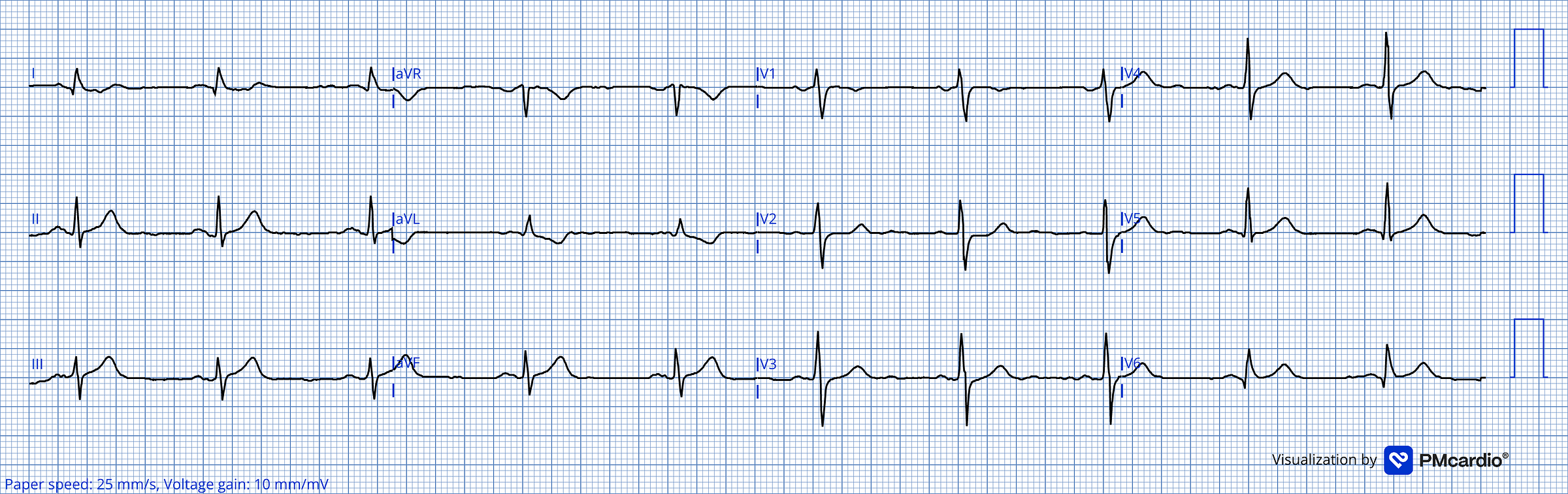
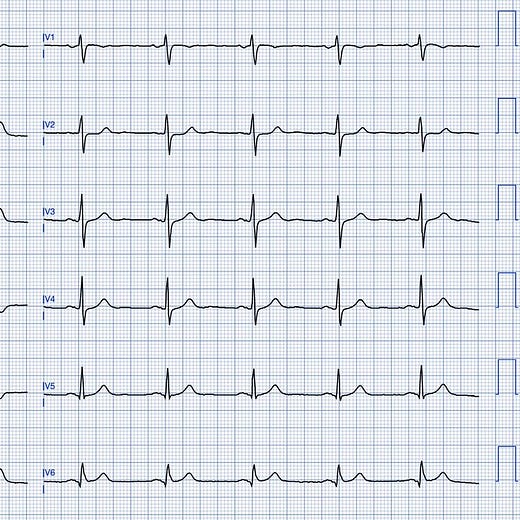
Here is the initial ECG recorded in the emergency department. What do you think?
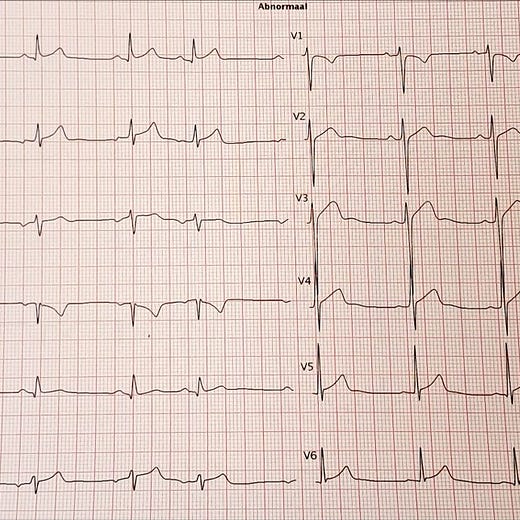
I shared this as a mini quiz on Twitter a couple of months ago (the ECG from today’s case is the one on the right, the other is from a different patient). If we exclude responses from regular followers and mentees of Dr. Smith’s ECG Blog, the answers were mostly incorrect. Test yourself here:
The machine interpretation of the initial, blue grid ECG above reads: Sinus rhythm, Normal electrical axis, QRS(T) contour deviation, Consider anterolateral myocardial injury.
Herman: These conventional, rule-based algorithms that print diagnostic statements on ECGs date back to the 1980s. If you are interested in how they work, here is the most recent physician guide for GE’s Marquette 12SL algorithm, which was available in this case.
The ED physicians at the non-PCI center over-read the ECG as “no obvious repolarization abnormality.” If you are only looking for tombstone ST elevation, this interpretation is understandable…
However, those T-waves in the inferior leads are enormous, almost the same size relative to the QRS complex. Broad, bulky, symmetric — what we would call hyperacute T-waves. There is almost no ST-elevation. Yet there is clear reciprocal change in leads I and aVL, along with relative ST depression maximal in V2–V3, where slight ST elevation would normally be expected (this supports posterior involvement).
Unfortunately, the ED physicians at the non-PCI center did not yet have access to the Queen of Hearts AI ECG Model (this case ultimately played a key role in convincing them). The initial high-sensitivity troponin I came back at 120 ng/L (URL 53). Repeat at 108 ng/L.
Smith: Do not be reassured by this. A flat or slightly falling troponin does not exclude acute coronary occlusion, especially in the setting of resolving or intermittent chest pain. This pattern is well described and frequently misinterpreted.
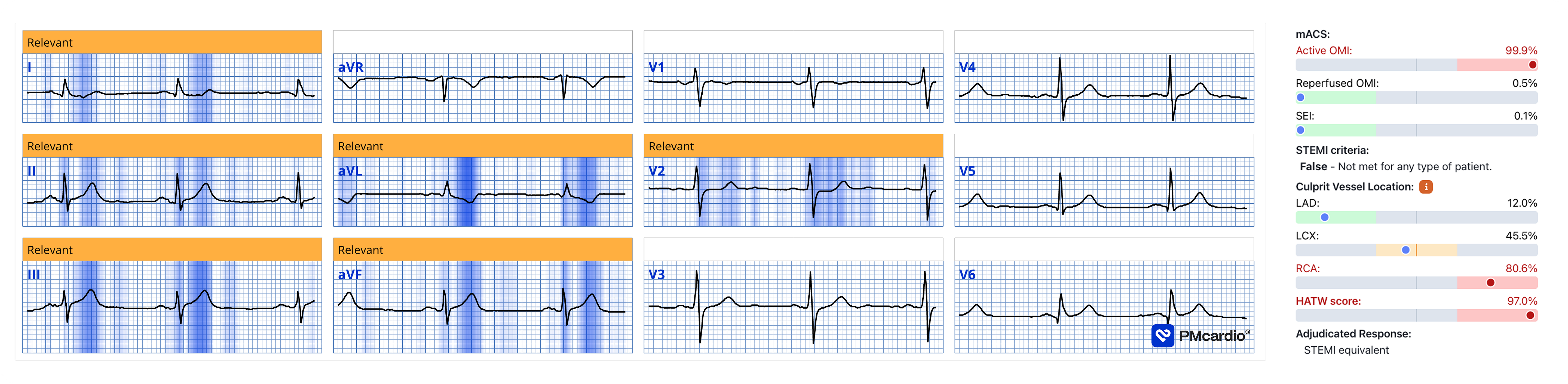
The team interpreted this as “not significantly rising” and did not activate the STEMI pathway. They administered pantoprazole 80 mg IV and aspirin 1 g IV and opted for observation. Hours later, the on-call cardiologist at the primary PCI center was contacted for an emergent NSTEMI consult. They had prospective access to the Queen of Hearts, and here is the AI ECG interpretation:
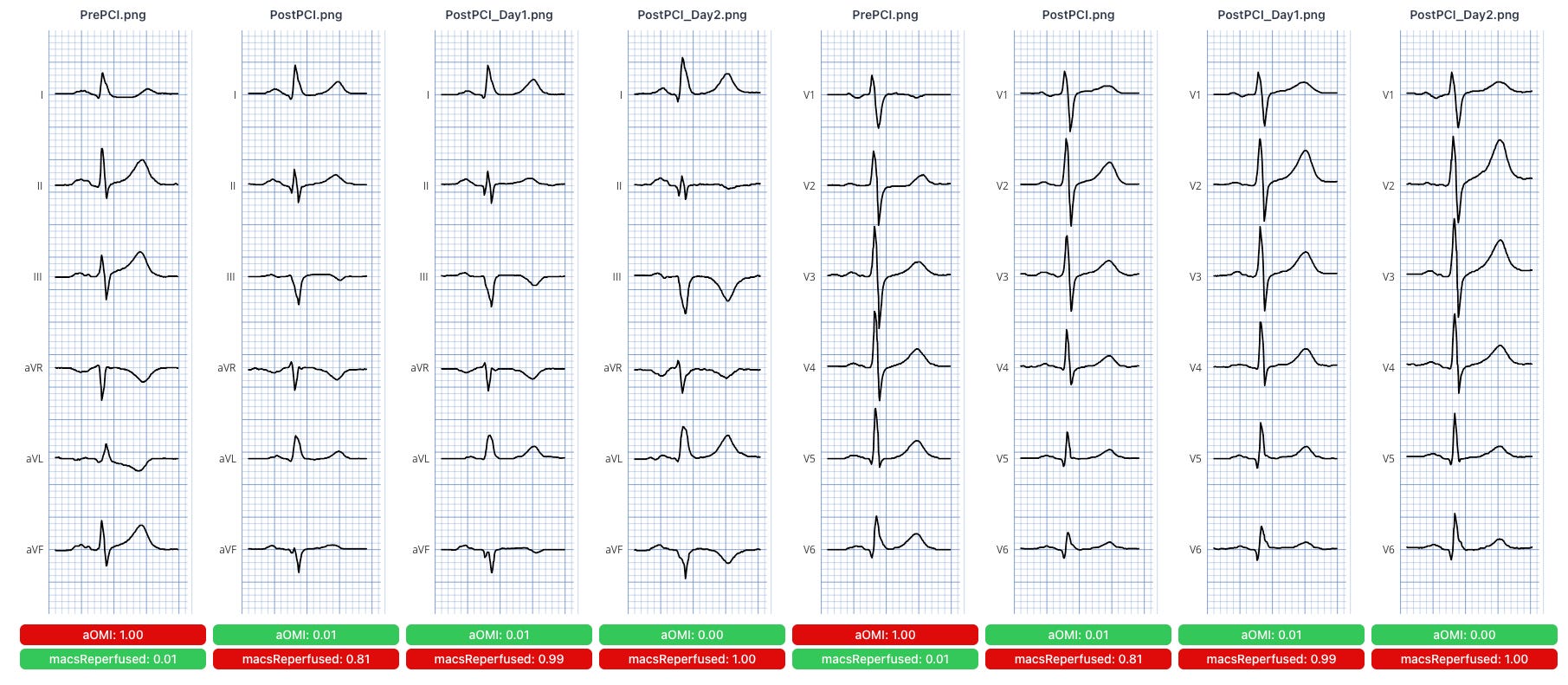
Queen of Hearts interpretation of the index ECG: the sidebar, an experimental feature, shows a 99.9% probability of active OMI. It also reports a HATW score of 97% and an 80.6% probability that the culprit lesion is in the RCA.
Herman: The AI models output values ranging from 0 to 1, with 0.5 as the threshold for positivity. Values close to 1 can be interpreted as confidently positive. A value of 99.9% represents the highest possible output, corresponding to an extremely low likelihood of false positives. In all-comer ED cohorts, false positives at this level are rare, approximately 1 in 100,000."
On the Cath Lab Table
Revascularization time: 10 AM (5 hours later).
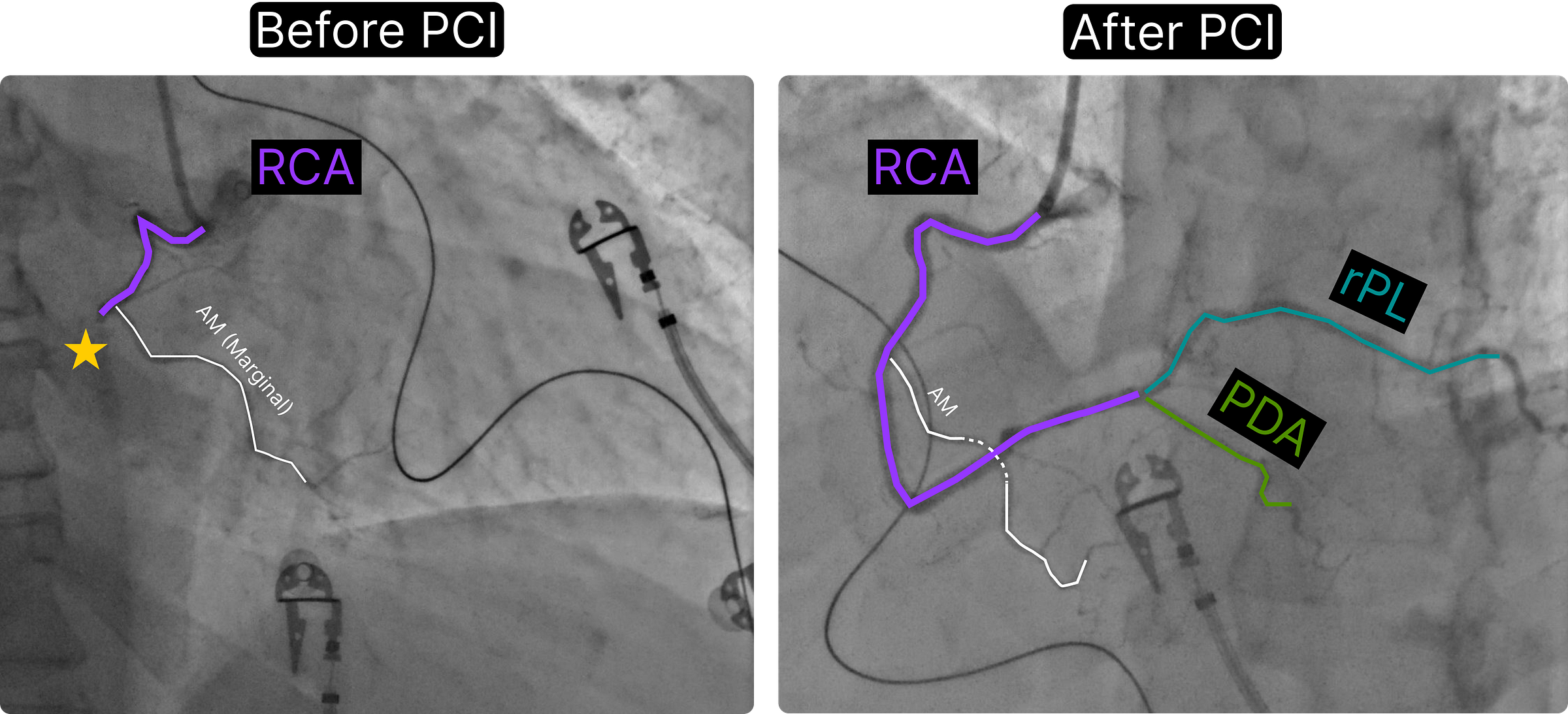
Coronary angiography: Right-dominant system. Mid-RCA: 100% occlusion (SYNTAX segment 2).
This was an acute infero-posterior wall myocardial infarction from total occlusion of the mid-right coronary artery.
The patient went into ventricular fibrillation on the cath lab table. Successful cardioversion. Hemodynamic stability restored. Angiographic post PCI result: excellent.
See below the annotated coronary angiogram before and after PCI, using the color scheme defined by (sadly now EP fellow) Dr. Willy Frick. If you are new to coronary angiography, I strongly recommend his introductory guide.
The third troponin (now measured as hs-cTnT with an upper reference limit of 14 ng/L), exceeded 1,800 ng/L. Unfortunately, serial measurements were not continued to peak, so the full extent of myocardial injury remains unknown.
Reperfusion T-wave Inversion Is Not Just an Anterior Phenomenon
Most clinicians learn about “Wellens syndrome” as deep, symmetric T-wave inversions in V2–V4 after chest pain — a pattern classically linked to the LAD and anterior territory, signaling a critical proximal lesion.
I have two problems with this framing. First, clinicians mainly associate it with the LAD — yet the same physiology operates in every territory (as you’ll see below). Second, “Wellens Type A” and “Wellens Type B” are not two distinct entities but two stages of the same pathophysiological process: the biphasic T-wave is earlier, the deep symmetric inversion is later. Calling them separate syndromes obscures what is actually happening.
Smith: The fact that “Wellens” represents coronary artery reperfusion (or spontaneous recanalization) on the ECG is apparent only to regular followers of my blog. It has never been formally described as such in the literature.
What is actually happening: after reperfusion of the culprit artery — whether LAD, RCA, or LCx — the T-waves in the corresponding distribution flatten and then invert as stunned myocardium recovers. This is a dynamic, time-dependent process. The correct name for it is simply reperfusion T-wave inversion, localized to the territory of the reopened artery.
In this case, focus on the inferior leads in the tracings below. The previously hyperacute, broad T-waves in II, III, and aVF — the signature of the occluded RCA — progressively flatten and then invert after successful PCI. The same pattern you would expect in V2–V4 after LAD reperfusion (Wellens). Different territory, identical physiology.
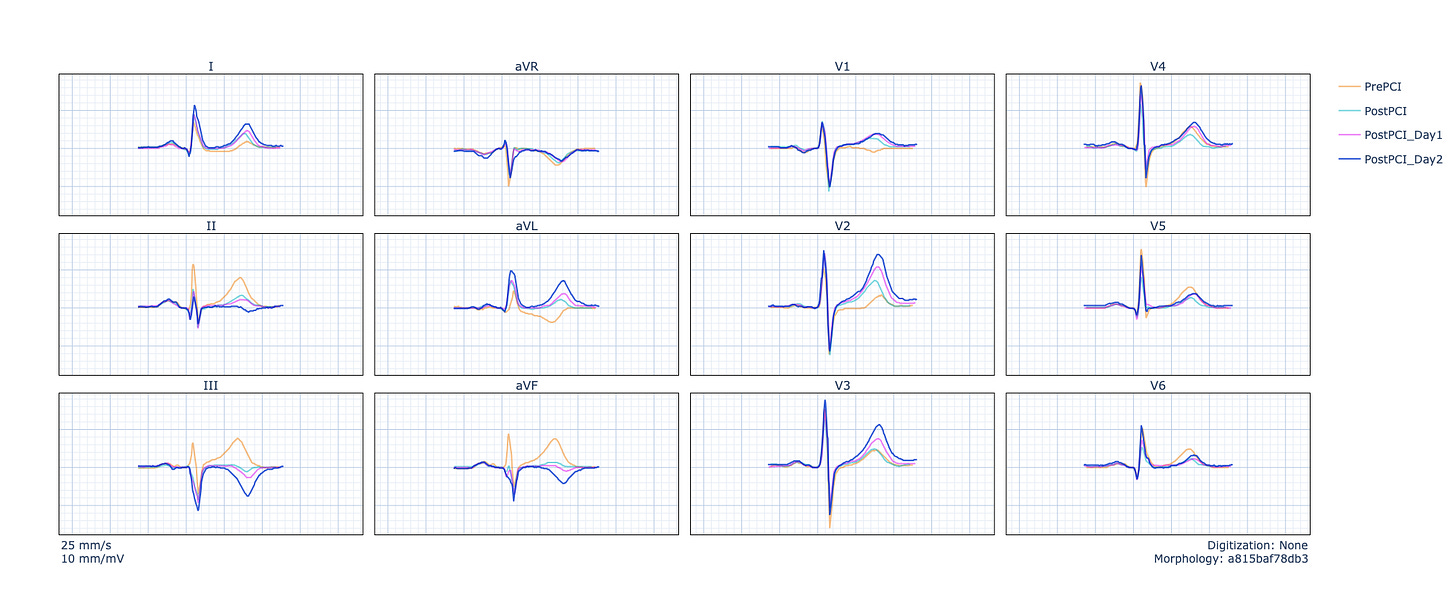
Why does recognizing this pattern matter? While T-wave inversion after PCI is an expected finding, the same pattern appears when patients present to the ED with an ECG that already shows reperfusion — meaning the artery opened spontaneously before arrival. The Queen of Hearts currently classifies these as “high-risk NSTEMI,” recognizing that the culprit artery is open at the time of the tracing but that the underlying lesion remains unstable and dangerous (and could re-occlude at any moment, since there was no mechanical intervention yet).
One critical distinction worth dwelling on — and one I hear get collapsed too often, even among clinicians who have embraced the OMI paradigm: the binary framing of “occluded vs. non-occluded” misses a third state that changes management entirely.
There are actually three clinically distinct scenarios:
Active OMI: the artery is occluded right now. The ECG shows ST-elevation or in 50% of cases, STEMI equivalents (such as hyperacute T-waves, minimal ST-elevation, or other active ischemic changes). This patient needs the cath lab immediately.
Reperfused OMI: the artery was occluded but has spontaneously recanalized before the ECG was recorded. The ECG now shows the reperfusion pattern: T-wave inversions in the culprit territory, often with a history of chest pain that has partially or fully resolved. Note: spontaneous recanalization (if the artery opens up on its own) cannot be differentiated from mechanical reperfusion (if the artery is opened up via PCI in the cath lab).
No OMI (includes NOMI and no MI): the artery was never occluded, or no myocardial infarction occurred. This category includes Type 2 MI, such as demand ischemia from another cause, as well as Type 1 MI due to non-occlusive plaque rupture, the “lucky” patients who never fully occlude. It also encompasses, by far, the largest group of chest pain patients, those without any myocardial infarction or troponin elevation.
Key Learning Points
Hyperacute T-waves are the earliest — and most commonly missed — sign of OMI. Very high specificity. No ST-elevation required. Look at the T/QRS ratio, the breadth, and the symmetry. Reciprocal changes seal the diagnosis. Read more about our HATW formula paper in JACC: Advances.
Two troponins that are “flat but elevated” do not exclude active occlusion. Waning chest pain with a plateau troponin is classic for stuttering OMI or spontaneous reperfusion — not reassurance.
Reperfusion T-wave inversions occur in every territory, not just anterior.
Prolonged ischemic time kills myocardium and creates procedural risk. This patient arrested on the table. Every hour of delay is both muscle lost and arrhythmia risk gained.
AI interpretation at the point of first contact can change outcomes. The Queen of Hearts read this ECG as 99.9% Active OMI. That information was not available at the referring site when it mattered most.





Cool case. A great example of recognizing the sharpest teeth of T.